Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
phenyldiazomethane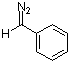  |
dichloromethane | N Param.: 9.35 sN Param.: 0.83 | Chem. Eur. J. 2003, 9, 4068-4076 10.1002/chem.200304913 | |
phenylsilane  |
dichloromethane | N Param.: 0.06 sN Param.: 0.71 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
phenylsulfinate (in 50W50AN)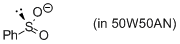  |
water-MeCN mix | N Param.: 13.75 sN Param.: 0.68 | J. Am. Chem. Soc. 2010, 132, 4796-4805 10.1021/ja9102056 | |
phenylsulfinate (in DMSO)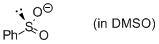  |
DMSO | N Param.: 19.60 sN Param.: 0.60 | J. Am. Chem. Soc. 2010, 132, 4796-4805 10.1021/ja9102056 | |
phenylsulfinate (in MeCN)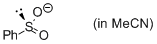  |
MeCN | N Param.: 20.11 sN Param.: 0.59 | J. Am. Chem. Soc. 2010, 132, 4796-4805 10.1021/ja9102056 | |
phosphaethynolate ion (in MeCN)  |
MeCN | N Param.: 19.02 sN Param.: 0.82 | Chem. Sci. 2024, 15, 14406-14414 10.1039/D4SC03518F | |
phthalimide anion (in DMSO)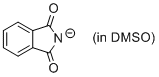  |
DMSO | N Param.: 15.52 sN Param.: 0.67 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
phthalimidoperoxyhexanoate (in H2O)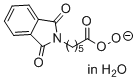  |
water | N Param.: 16.02 sN Param.: 0.54 | Angew. Chem. Int. Ed. 2017, 56, 13279-13282 10.1002/anie.201707086 | |
piperazine (in water)  |
water | N Param.: 17.22 sN Param.: 0.50 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
piperidin-1-yl dithiocarbamate (in MeCN)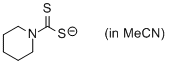  |
MeCN | N Param.: 23.84 sN Param.: 0.57 | Org. Biomol. Chem. 2011, 9, 8046-8050 10.1039/c1ob06245j | |
piperidine (in 91M9AN)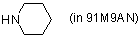  |
MeOH-MeCN mix | N Param.: 15.63 sN Param.: 0.64 | Angew. Chem. Int. Ed. 2006, 45, 3869-3874 10.1002/anie.200600542 | |
piperidine (in DMSO)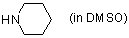  |
DMSO | N Param.: 17.19 sN Param.: 0.71 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
piperidine (in MeCN)  |
MeCN | N Param.: 17.35 sN Param.: 0.68 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
piperidine (in water)  |
water | N Param.: 18.13 sN Param.: 0.44 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
potassium (1-(tert-butoxycarbonyl)-1H-indol-2-yl)trifluoroborate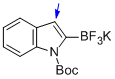  |
MeCN | N Param.: 6.46 sN Param.: 0.96 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium (1-(tert-butoxycarbonyl)-1H-indol-3-yl)trifluoroborate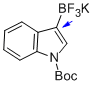  |
MeCN | N Param.: 4.06 sN Param.: 0.79 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium (1-(tert-butoxycarbonyl)-5-methoxy-1H-indol-2-yl)trifluoroborate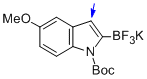  |
MeCN | N Param.: 7.10 sN Param.: 1.18 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium allyltrifluoroborate (in MeCN)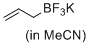  |
MeCN | N Param.: 5.29 sN Param.: 0.87 | J. Am. Chem. Soc. 2017, 139, 15324-15327 10.1021/jacs.7b10240 | |
potassium indenide (in DMSO)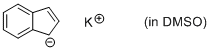  |
DMSO | N Param.: 24.16 sN Param.: 0.68 | Angew. Chem. Int. Ed. 2015, 54, 12497-12500 10.1002/anie.201501385 | |
potassium trifluoro(1-methyl-1H-indol-2-yl)borate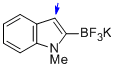  |
MeCN | N Param.: 9.55 sN Param.: 1.16 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium trifluoro(1-methyl-1H-indol-5-yl)borate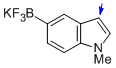  |
MeCN | N Param.: 8.77 sN Param.: 1.09 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium trifluoro(3-methoxy-thiophen-2-yl)borate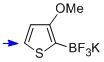  |
MeCN | N Param.: 7.32 sN Param.: 0.90 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium trifluoro(furan-3-yl)borate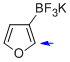  |
MeCN | N Param.: 6.83 sN Param.: 0.93 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
prenyl-tributylstannane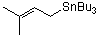  |
dichloromethane | N Param.: 4.74 sN Param.: 1.15 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
prolinate (in MeCN)  |
MeCN | N Param.: 19.95 sN Param.: 0.68 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
proline (anionic, in water)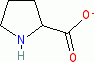  |
water | N Param.: 18.08 sN Param.: 0.50 | Org. Biomol. Chem. 2007, 5, 3814-3820 10.1039/b713778h | |
propargylamine (in water)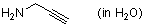  |
water | N Param.: 12.29 sN Param.: 0.59 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
propene  |
dichloromethane | N Param.: -2.41 sN Param.: 1.00 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
purine anion (in DMSO)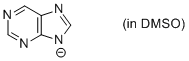  |
DMSO | N Param.: 15.03 sN Param.: 0.77 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
purine anion (in water)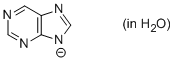  |
water | N Param.: 11.00 sN Param.: 0.54 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
pyridin-4-yl((4-(trifluoromethyl)phenyl)sulfonyl)amide (Ph4P+) (in MeCN)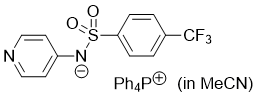  |
MeCN | N Param.: 19.51 sN Param.: 0.47 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4N+) (in MeCN)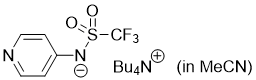  |
MeCN | N Param.: 16.68 sN Param.: 0.59 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4P+) (in MeCN)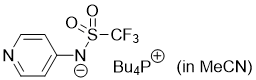  |
MeCN | N Param.: 16.48 sN Param.: 0.60 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Ph4P+) (in CH2Cl2)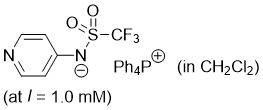  |
dichloromethane | N Param.: 17.88 sN Param.: 0.73 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Ph4P+) (in MeCN)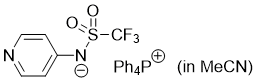  |
MeCN | N Param.: 16.38 sN Param.: 0.60 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
pyridin-4-yl(tosyl)amide (Ph4P+) (in MeCN)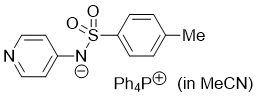  |
MeCN | N Param.: 17.19 sN Param.: 0.64 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
pyridine (in CH2Cl2)  |
dichloromethane | N Param.: 12.90 sN Param.: 0.67 | Chem. Eur. J. 2007, 13, 336-345 10.1002/chem.200600941 | |
pyridine (in H2O)  |
water | N Param.: 11.05 sN Param.: 0.73 | Chem. Eur. J. 2007, 13, 336-345 10.1002/chem.200600941 | |
pyridine (in MeCN)  |
MeCN | N Param.: 13.60 sN Param.: 0.60 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
pyrrole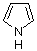  |
dichloromethane | N Param.: 4.63 sN Param.: 1.00 | Chem. Eur. J. 2003, 9, 2209-2218 10.1002/chem.200204666 | |
pyrrolidin-1-yl dithiocarbamate (in MeCN)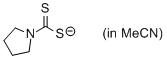  |
MeCN | N Param.: 22.40 sN Param.: 0.63 | Org. Biomol. Chem. 2011, 9, 8046-8050 10.1039/c1ob06245j | |
pyrrolidine (in 91M9AN)  |
MeOH-MeCN mix | N Param.: 15.97 sN Param.: 0.63 | Angew. Chem. Int. Ed. 2006, 45, 3869-3874 10.1002/anie.200600542 | |
pyrrolidine (in water)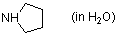  |
water | N Param.: 17.21 sN Param.: 0.49 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
pyrrolidine (inMeCN)  |
MeCN | N Param.: 18.58 sN Param.: 0.61 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
quinidine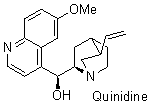  |
dichloromethane | N Param.: 10.54 sN Param.: 0.74 | J. Org. Chem. 2009, 74, 7157-7164 10.1021/jo901670w | |
quinine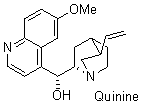  |
dichloromethane | N Param.: 10.46 sN Param.: 0.75 | J. Org. Chem. 2009, 74, 7157-7164 10.1021/jo901670w | |
quinuclidine (in MeCN)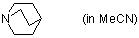  |
MeCN | N Param.: 20.54 sN Param.: 0.60 | Angew. Chem. Int. Ed. 2007, 46, 6176-6179 10.1002/anie.200701489 | |
saccharin anion (in MeCN)  |
MeCN | N Param.: 10.78 sN Param.: 0.89 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
Se-BTM (in CH2Cl2)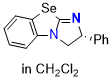  |
dichloromethane | N Param.: 14.27 sN Param.: 0.63 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-DHPB (in CH2Cl2)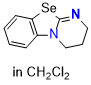  |
dichloromethane | N Param.: 15.16 sN Param.: 0.66 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-HBTM (in CH2Cl2)  |
dichloromethane | N Param.: 14.42 sN Param.: 0.67 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-HyperBTM (in DCM)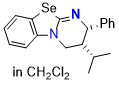  |
dichloromethane | N Param.: 16.11 sN Param.: 0.58 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-HyperBTM (in MeCN)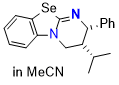  |
MeCN | N Param.: 15.84 sN Param.: 0.57 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-HyperBTM (in THF)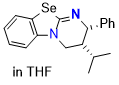  |
THF | N Param.: 14.19 sN Param.: 0.77 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-tetramisole (Se-TM) (in CH2Cl2)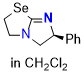  |
dichloromethane | N Param.: 15.26 sN Param.: 0.60 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
semicarbazide (in water)  |
water | N Param.: 11.05 sN Param.: 0.52 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
serine (anionic, in water)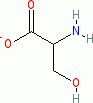  |
water | N Param.: 13.16 sN Param.: 0.55 | Org. Biomol. Chem. 2007, 5, 3814-3820 10.1039/b713778h | |
SO3(2-) (in water)  |
water | N Param.: 16.83 sN Param.: 0.56 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
sodium indenide (in DMSO)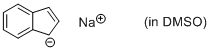  |
DMSO | N Param.: 23.74 sN Param.: 0.71 | Angew. Chem. Int. Ed. 2015, 54, 12497-12500 10.1002/anie.201501385 | |
styrene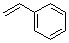  |
dichloromethane | N Param.: 0.78 sN Param.: 0.95 | J. Am. Chem. Soc. 2001, 123, 9500-9512 10.1021/ja010890y | |
succinimide anion (in DMSO)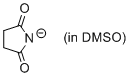  |
DMSO | N Param.: 16.03 sN Param.: 0.66 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
t-butylperoxy anion (in H2O)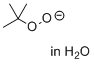  |
water | N Param.: 14.29 sN Param.: 0.51 | Angew. Chem. Int. Ed. 2017, 56, 13279-13282 10.1002/anie.201707086 | |
TCAP (in MeCN)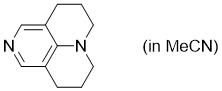  |
MeCN | N Param.: 15.60 sN Param.: 0.68 | Chem. Eur. J. 2013, 19, 6435-6442 10.1002/chem.201204452 | |
Te-BTM (in CH2Cl2)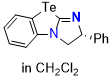  |
dichloromethane | N Param.: 14.62 sN Param.: 0.66 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Te-DHPB (in CH2Cl2)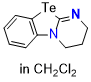  |
dichloromethane | N Param.: 16.63 sN Param.: 0.65 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Te-HyperBTM (in DCM)  |
dichloromethane | N Param.: 16.63 sN Param.: 0.63 | ChemistryEurope 2026, 4, e202500443 10.1002/ceur.202500443 | |
tert-butanol (in MeCN)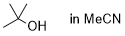  |
MeCN | N Param.: 5.35 sN Param.: 0.72 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
tert-butyl hydrazinecarboxylate (in MeCN)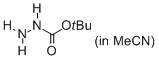  |
MeCN | N Param.: 11.40 sN Param.: 0.70 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
tert-butyl indole-1-carboxylate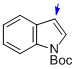  |
MeCN | N Param.: 1.68 sN Param.: 1.26 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
tert-butyl isocyanide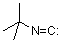  |
dichloromethane | N Param.: 5.47 sN Param.: 0.77 | Angew. Chem. Int. Ed. 2007, 46, 3563-3566 10.1002/anie.200605205 | |
tert-butylamine (in MeCN)  |
MeCN | N Param.: 12.35 sN Param.: 0.72 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
tert-butylamine (in water)  |
water | N Param.: 10.48 sN Param.: 0.65 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
tetra-sec.butylstannane (in 1,2-DCE)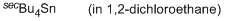  |
1,2-dichloroethane | N Param.: -1.10 sN Param.: 1.15 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetrabutylstannane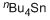  |
dichloromethane | N Param.: -0.30 sN Param.: 1.07 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetraethylgermane (in MeCN)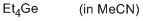  |
MeCN | N Param.: -4.70 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetraethylplumbane (in MeCN)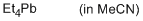  |
MeCN | N Param.: 0.10 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetraethylsilane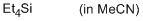  |
MeCN | N Param.: -6.50 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetraethylstannane (in 1,2-DCE)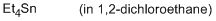  |
1,2-dichloroethane | N Param.: -1.90 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetraethylstannane (in MeCN)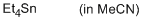  |
MeCN | N Param.: -2.40 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetrahydroborate ion (in water)  |
water | N Param.: 12.10 sN Param.: 0.79 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetrahydrofuran (in dichloromethane)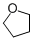  |
dichloromethane | N Param.: -3.50 sN Param.: 0.80 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetrahydrothiophene (in CH2Cl2)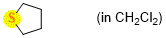  |
dichloromethane | N Param.: 13.10 sN Param.: 0.72 | Chem. Eur. J. 2021, 21, 11367-11376 10.1002/chem.202100977 | |
tetrahydrothiophene (in MeCN)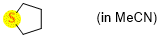  |
MeCN | N Param.: 13.30 sN Param.: 0.72 | Chem. Eur. J. 2021, 21, 11367-11376 10.1002/chem.202100977 | |
tetrahydrothiopyran (in CH2Cl2)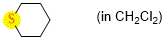  |
dichloromethane | N Param.: 11.94 sN Param.: 0.75 | Chem. Eur. J. 2021, 21, 11367-11376 10.1002/chem.202100977 | |
tetraisobutylstannane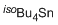  |
dichloromethane | N Param.: 1.35 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
tetrakis(5-methyl-furan-2-yl)borate  |
MeCN | N Param.: 9.09 sN Param.: 1.12 | Chem. Sci. 2012, 3, 878-882 10.1039/c2sc00883a | |
tetramethylethylene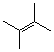  |
dichloromethane | N Param.: -1.00 sN Param.: 1.40 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
tetramisole (TM) (in CH2Cl2)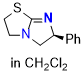  |
dichloromethane | N Param.: 13.86 sN Param.: 0.68 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
tetrapropylstannane (in 1,2-DCE)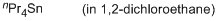  |
1,2-dichloroethane | N Param.: -0.50 sN Param.: 1.13 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
theophylline anion (in DMSO)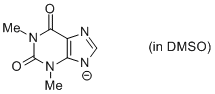  |
DMSO | N Param.: 14.78 sN Param.: 0.71 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
theophylline anion (in water)  |
water | N Param.: 10.06 sN Param.: 0.71 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
thioacetate (in MeCN)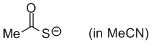  |
MeCN | N Param.: 21.20 sN Param.: 0.63 | Org. Biomol. Chem. 2011, 9, 8046-8050 10.1039/c1ob06245j | |
thiocyanate (in MeCN)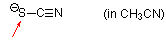  |
MeCN | N Param.: 17.94 sN Param.: 0.60 | J. Am. Chem. Soc. 2003, 125, 14126-14132 10.1021/ja037317u | |
thiocyanate (in MeCN)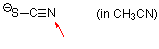  |
MeCN | N Param.: 12.13 sN Param.: 0.60 | J. Am. Chem. Soc. 2003, 125, 14126-14132 10.1021/ja037317u | |
thiophene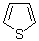  |
dichloromethane | N Param.: -1.01 sN Param.: 1.10 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
thiophenolate (in DMSO)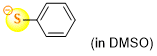  |
DMSO | N Param.: 23.36 sN Param.: 0.74 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
threonine (anionic, in water)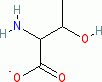  |
water | N Param.: 12.69 sN Param.: 0.60 | Org. Biomol. Chem. 2007, 5, 3814-3820 10.1039/b713778h | |
THTP (3,5,6,7-tetrahydro-2H-thiazolo[3,2-a]pyrimidine)  |
dichloromethane | N Param.: 14.45 sN Param.: 0.78 | J. Org. Chem. 2011, 76, 5104-5112 10.1021/jo200803x | |
thymine anion (in DMSO)  |
DMSO | N Param.: 17.63 sN Param.: 0.62 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
thymine anion (in water)  |
water | N Param.: 11.17 sN Param.: 0.51 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 |
