 Theoretical Chemistry Group - Faculty for Chemistry and Pharmacy
Theoretical Chemistry Group - Faculty for Chemistry and Pharmacy
 Theoretical Chemistry Group - Faculty for Chemistry and Pharmacy
Theoretical Chemistry Group - Faculty for Chemistry and Pharmacy
| Molecular structures of Vitamin K2 | |
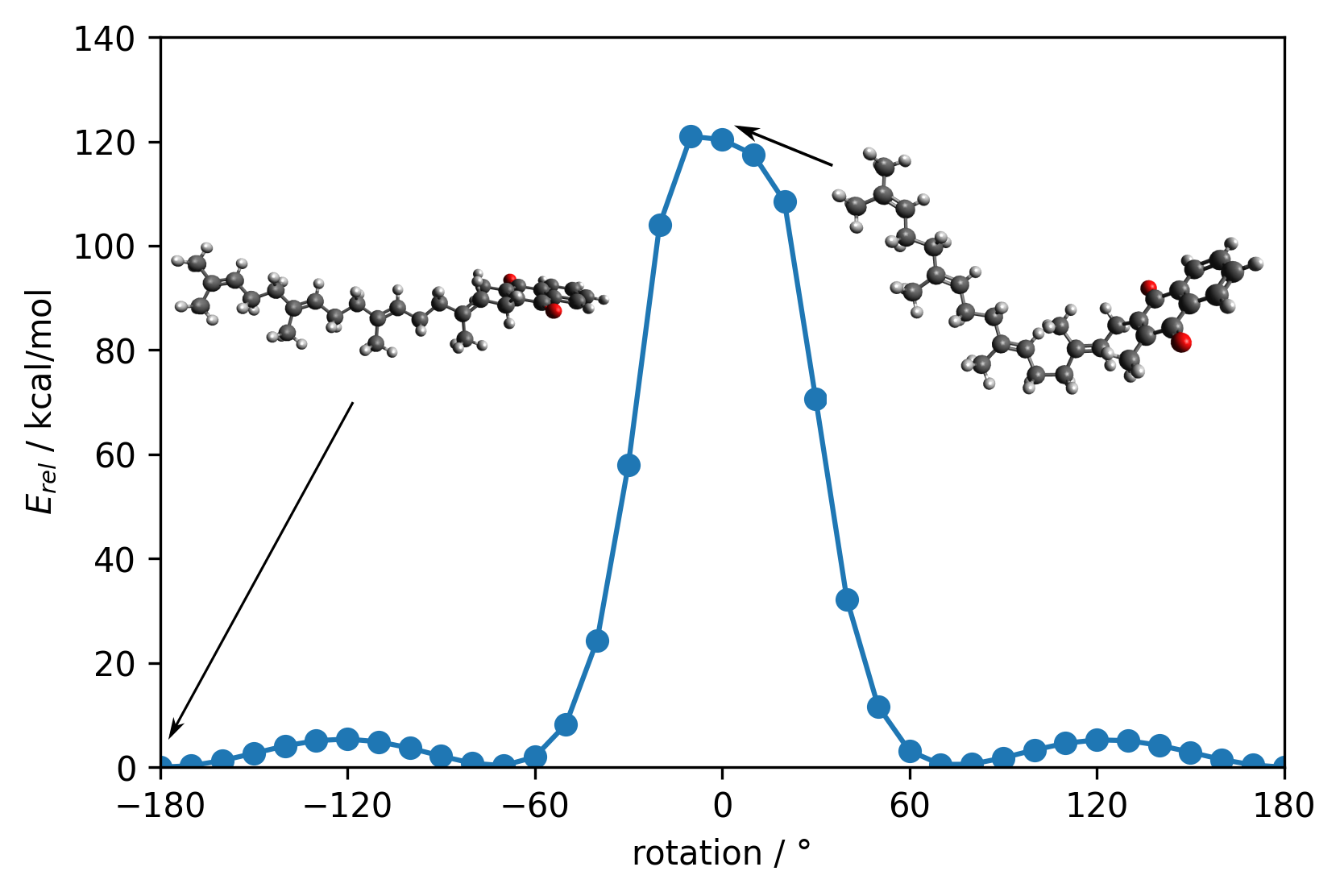 |
Description / Information Structures. |
|
If used for publication, please cite the following reference: F. H. Bangerter, M. Glasbrenner, C. Ochsenfeld, Download vitk2_tar.gz (36 kB) |
|
| Additional Data of the NAMD Simulations (2) | |
 |
Description / Information Structures, initial conditions, and movies. |
|
If used for publication, please cite the following reference: Download namd2_tar.gz (7 MB) |
|
| Structure of Sirtuin 5 | |
 |
Description / Information PDB file of Sirtuin 5. |
|
If used for publication, please cite the following reference: Download sirt5.pdb (5 MB) |
|
| Additional Data of the NAMD Simulations | |
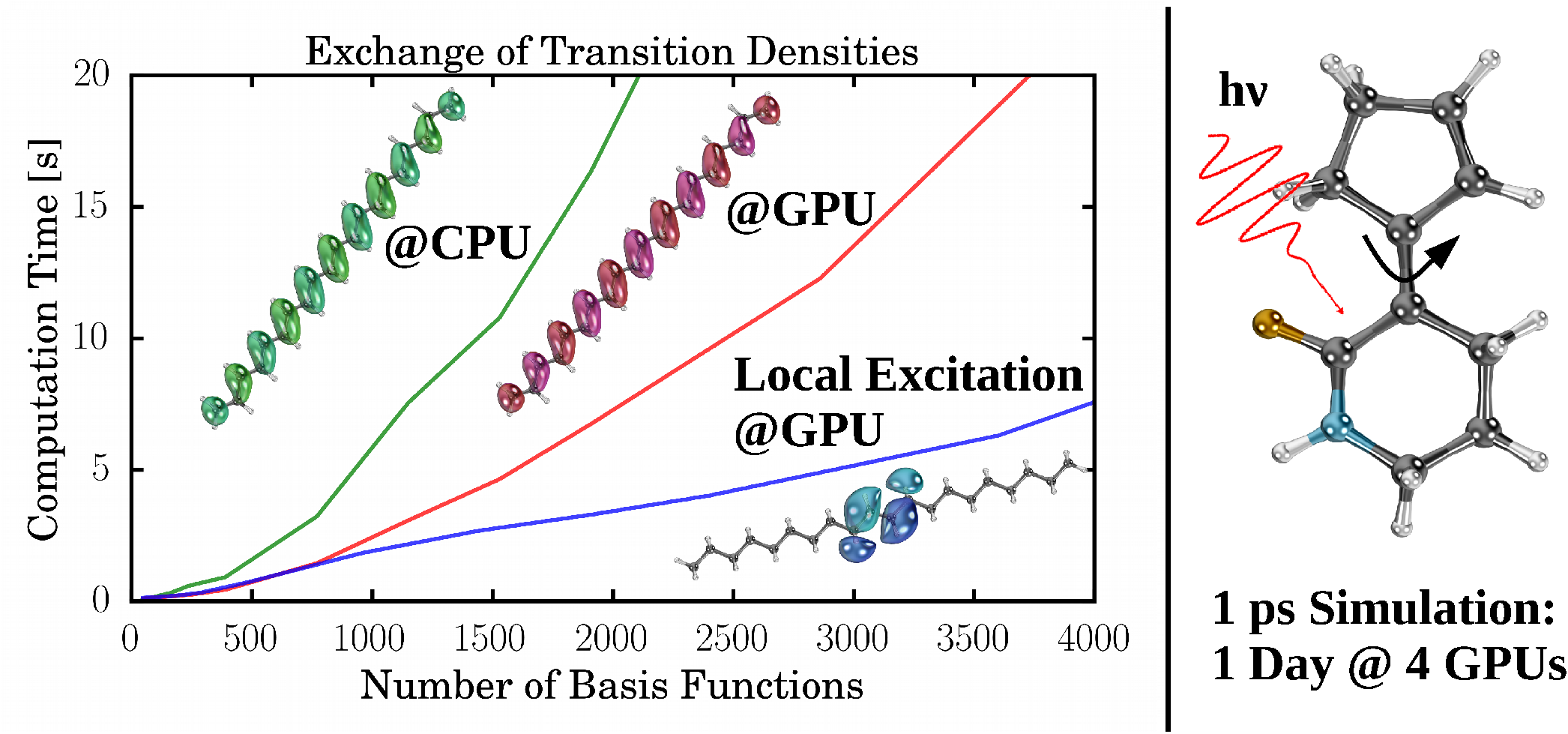 |
Description / Information Movies, initial conditions, and energy plots of the NAMD simulations. Molecular structures used for the timings. |
|
If used for publication, please cite the following reference: L. D. M. Peters, J. Kussmann, C. Ochsenfeld, J. Chem. Theory Comput., 15, 6647-6659 (2019). Download namd_tar.gz (134 MB) |
|
| Tutorial and Scripts for identifying free energy hot-spots | |
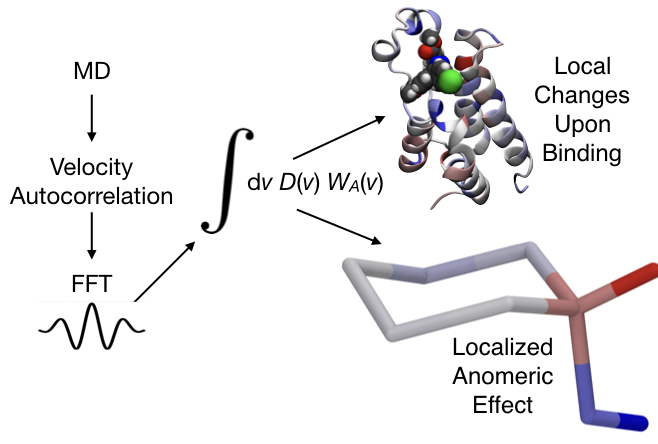 |
Description / Information An hands-on tutorial for identifying free energy hot-spots in molecular transformations, including scripts and an example. |
|
If used for publication, please cite the following reference: Download hot-spots_tar.gz (271 kB) |
|
| Molecular structures for g-tensor calculations | |
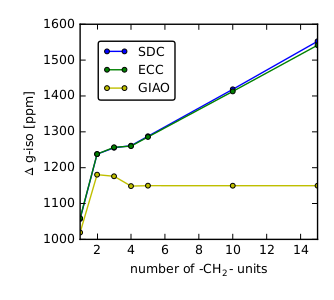 |
Description / Information Molecular structures that were used to benchmark the influence of the gauge origin on the electronic g-tensor. |
|
If used for publication, please cite the following reference: M. Glasbrenner, S. Vogler, C. Ochsenfeld, J. Chem. Phys. 150, 024101 (2018). Download g-tensor.tar.gz (48 kB) |
|
| Molecular structures for preselective screening in exact exchange gradient calculations | |
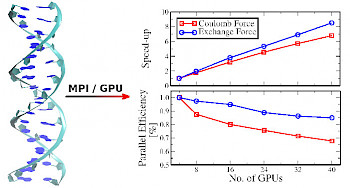 |
Description / Information Molecular structures that were used to analyze the impact of preselective screening in O(N) exact exchange gradient calculations. |
|
If used for publication, please cite the following reference: J. Kussmann and C. Ochsenfeld, J. Chem. Theory Comput. 11, 918-922 (2015). Download prelink_grad_structures.tgz (39 kB) |
|
| Molecular structures for benchmarking heteronuclear NMR shifts | |
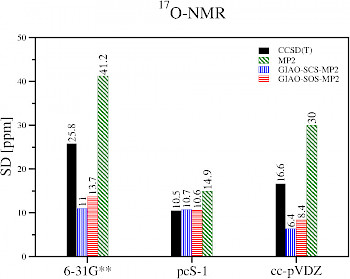 |
Description / Information Molecular test sets for 19F-, 17O-, 31P- and 15N-NMR shifts. The structures were optimized at the CCSD(T)/cc-pVTZ level (see publication for further details).
|
|
If used for publication, please cite the following reference: M. Maurer and C. Ochsenfeld, J. Chem. Theory Comput. 11, 37 (2015) Download nmr-benchmark-set-f-n-o-p.tgz (4 kB) |
|
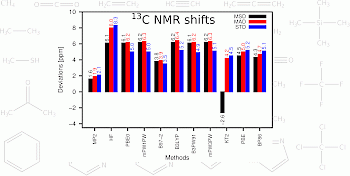 |
Description / Information A molecular test set covering a broad range of the NMR scale for hydrogen and carbon shifts with high relevance to organic chemistry. The global minimum structures were obtained at the CCSD(T)/cc-pVTZ level (see publication for further details). |
|
If used for publication, please cite the following reference: Download nmr-benchmark-set.tgz (3 kB) |
|
| Benchmark set for integral screening methods | |
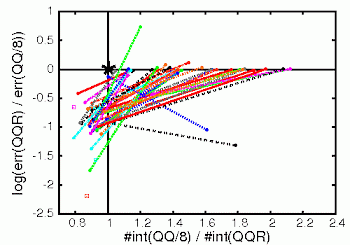 |
Description / Information A set of structures for benchmark/testing of integral screening methods and other approximations for large systems. The set contains insulating, delocalized, radical, and ionic systems with medium to very large systems up to 1707 atoms. |
|
If used for publication, please cite the following reference: Reference data is presented in the corresponding supplementary material. Download benchmark_set.tgz (339 kB) |
|
| Structures of oligoprolines | |
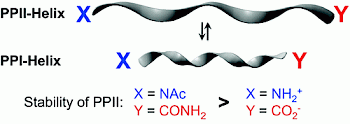 |
Description / Information HF/6-31G** optimized capped oligoprolines AcN−[Pro]15−CONH2 and AcN−[Pro]15−CO2CH3 for PP1 and PP2 helical arrangement of the backbone. The initial structures were generated using the program package Maestro assuming idealized dihedral angles for the respective helix backbone |
|
If used for publication, please cite the following reference: Download pp_structures.tgz (112 kB) |
|
| Structures of amylose chains | |
 |
Description / Information Structures of amylose chains containing n=1,2,4,8,16,32,48,64 -D-glucose units. The original structures have been obtained by the SWEET tool [A. Bohne, E. Lang, and. C. W. von der Lieth, J. Mol. Model. 4, 33 (1998); A. Bohne, E. Lang, and. C. W. von der Lieth, Bioinformatics 15, 767 (1999)]. Download
|
| Structures of Linear Alkanes and Glycine Chains |
|
Structures of linear n-alkanes CnH2n+2 with n=10,20,40,80,160. (9 kB) Structures of glycine chains Glyn with n=1,2,3,4,5,6,7,8,9,10,20,30,40,50,60,70,80,90. (35 kB) |
| SparseCholesky | |
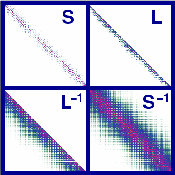 |
Description / Information SparseCholesky provides a set of routines for decomposing and inverting sparse, symmetric positive-definite matrices in a linear scaling fashion. In addition, a linear equation solver is supplied. In the sparse algebra routines the compressed sparse row (CSR) storage format is employed. The code is written in C/C++, but can easily embedded in Fortran code. If used for publication, please cite the following reference: S. Schweizer, J. Kussmann, B. Doser, and C. Ochsenfeld, J. Comp. Chem. 29, 1004 (2008). Download |
| DNA adenine-thymine fragments | |
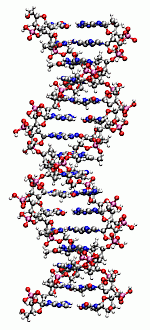 |
Description / Information Structures of DNAn fragments containing n=1,2,4,8,16 adenine-thymine base pairs. The original structures have been obtained by the Spartan package. If used for publication, please cite the following reference: B. Doser, D. S. Lambrecht, J. Kussmann, and C. Ochsenfeld, J. Chem. Phys. 130, 064107 (2009). Download |
| Modified Polyalanine Helices | |
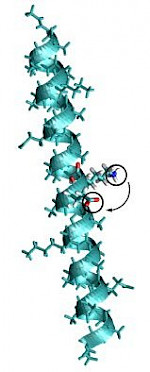 |
Description / Information Structures of model peptides consisting of a modified polyalanine helix in different protonation states. The original structures have been obtained by the Maestro package . If used for publication, please cite the following reference: C. V. Sumowski and C. Ochsenfeld, J. Phys. Chem. A 113 , 11734 (2009). Download |