 Group of Pablo Docampo - Faculty for Chemistry and Pharmacy
Group of Pablo Docampo - Faculty for Chemistry and Pharmacy
 Group of Pablo Docampo - Faculty for Chemistry and Pharmacy
Group of Pablo Docampo - Faculty for Chemistry and Pharmacy
Solar cells based on perovskite absorbers are the fastest growing photovoltaic technology in history. Since their introduction in early 2008 with a starting point power conversion efficiency of ~4%, they have now since left behind amorphous Silicon and are already achieving efficiencies of over 22%, competing with industry staples such as CIGS and polycrystalline Silicon. Perovskite solar cells have the potential to become a serious contender for the photovoltaic market due to their already demonstrated high device efficiencies, the low-cost of their starting materials and their compatibility with large-scale, solution processing techniques such as roll-to-roll printing on flexible substrates.
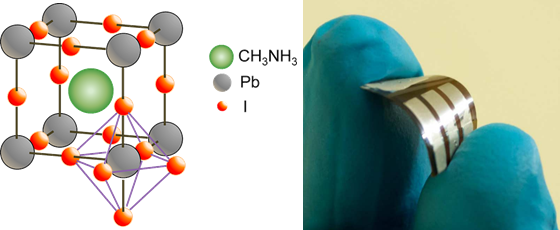
Docampo, P., Ball, J. M., Darwich, M., Eperon, G. E. & Snaith, H. J. Efficient organometal trihalide perovskite planar-heterojunction solar cells on flexible polymer substrates. Nature communications 4 (2013).
In our group, we have focused our research on the following topics:
A recent review article from our group gives an overview of the field (Adv. Energy Mater. 2017) The typical device structure prepared in our lab is shown in the scheme below.
Most perovskite-based solar cells contain lead, which is known to be toxic. Besides the health issues, also international laws force the recycling of electronic waste after their operational lifetime. We published a procedure to remove every layer of the solar cells separately, which gives the possibility to selectively isolate the different materials. In this way the toxic lead can be isolated and reused for making new solar cells. Also the expensive conductive glass (FTO) can be reused several times without losing the efficiency of the solar cells. With our simple recycling procedure, we address both the risk of contamination and the waste disposal of perovskite based solar cells while further reducing the cost of the system. This brings perovskite solar cells one step closer to their introduction into commercial systems.
Most perovskite based solar cells make use an organic hole transporting material, termed Spiro-OMeTAD. Even though this material is fully organic, the expensive starting materials, transition metal catalysts and inert reaction conditions required for the synthesis and extensive product purification make its production cost prohibitive.
We develop low cost alternatives for Spiro-OMeTAD, using simple and clean chemistry. We synthesized a hole transporting material (EDOT-OMeTPA) using Schiff-base condensation chemistry, in which water is the only by-product, making purification very straight forword. EDOT-OMeTPA showed comparable devices performance to state-of-the-art materials, but at a cost which is two orders of magnitude lower.
A Low Cost Azomethine-Based Hole Transporting Material for Perovskite Photovoltaics J. Mater. Chem. A, (2015), 3, 12159
Design Rules for the Preparation of Low-Cost Hole Transporting Materials for Perovskite Solar Cells with Moisture Barrier Properties, J. Mater. Chem. A, (2017), 5, 25200
We report a new synthesis approach for the fabrication of bromide-based perovskite planar films based on the control of the deposition environment. We achieve dense layers with large and perfectly oriented crystallites 5–10 μm in size. We additionally correlate photocurrent and perovskite crystal properties in photovoltaic devices and find that this parameter is maximized for ordered systems, with internal quantum efficiencies approaching unity.
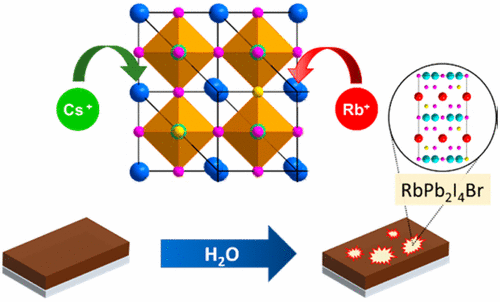
Rubidium and cesium cations have been recently identified as enhancers for perovskite solar cell performance. However, the impact of these inorganic cations on the stability of the perovskite crystal lattice has not been fully understood yet. We show that during the perovskite film synthesis, an unwanted side phase ( RbPb(I1–xBrx)3) is formed, while exposure to humid air leads to the rapid formation of another hitherto unreported side phase (RbPb2I4Br). The formation of the Rb-rich side phases not only results in a loss of light absorption but also extracts bromide ions from the photoactive perovskite phase, thereby reducing its band gap. In comparison, the moisture-assisted formation of a CsPb2I4Br phase upon Cs-addition occurs on a significantly longer time scale than its Rb analog. While the incorporation of Cs+ remains attractive for high-performance solar cells, the severe moisture-sensitivity of Rb-containing mixed-halide perovskites may create additional engineering challenges.
In collaboration with the University of Cambridge, we have developed novel light emitting devices based on perovskite structures. The materials prepared in our group exhibit high photoluminescence quantum yields, which make their application into lighting devices readily achievable.
Bright light-emitting diodes based on organometal halide perovskite. Nature Nanotechnology (2014).