Development of non-viral gene transfer vectors
Introduction
At present a deciding factor for progress in somatic gene therapy is the development of efficient and safe delivery systems. While several efficient vector systems (viral and nonviral) are available for ex vivo gene therapy, in vivo gene therapy is strongly hampered by the lack of efficient and target-specific vectors. However, a series of therapeutic applications can only be approached by in situ modification of patient cells, and also from the practical and economic point, in vivo gene transfer seems much more attractive (no laborious ex vivo cell culture). Our efforts are focused on the improvement of nonviral gene transfer systems (previously established in our lab) for in vivo gene therapy (see also insert 1 ). DNA complexes are characterised in regard to physical parameters (high solubility, small size, low charge) as well as minimum unspecific interactions with body fluids, tissues or cells (e.g. minimum inhibition by blood components).
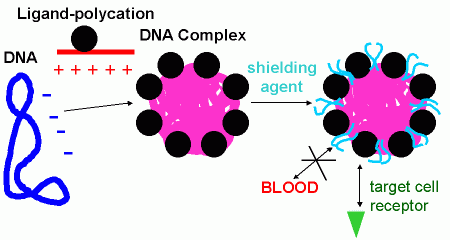
Intracellular barriers
Despite their ability to transfer plasmid DNA into a variety of cell lines in vitro and to a limited number of cell types in vivo, surprisingly little is known about the mode of entry and intracellular trafficking of non-viral gene transfer particles (see figure below). Uptake into cells seems to be no limiting step, but cytoplasmic release and entry of DNA into the nucleus are bottlenecks in gene transfer. Nuclear membrane breakdown during cell division may allow the passive inclusion of DNA (complexes) into the nuclear compartment. This may be one reason why actively dividing cells are easier to transfect than resting cells.
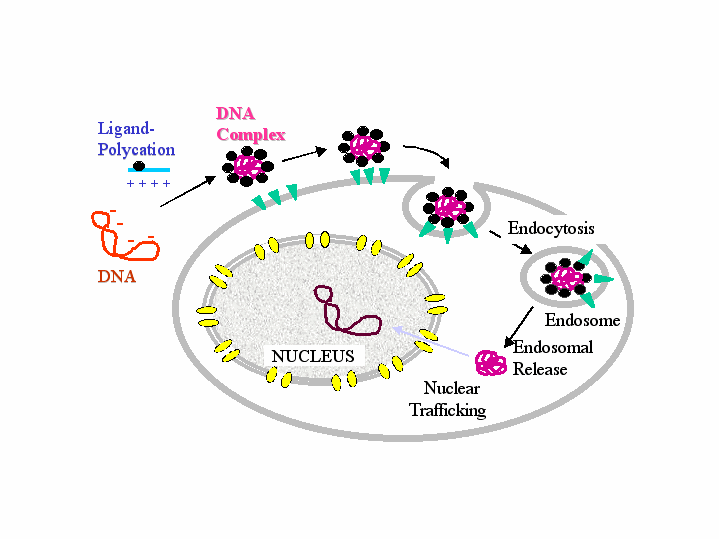
Reports suggested that some polycations and also nuclear localisation sequences may facilitate the nuclear uptake of DNA complexes. We are currently investigating the possibility to incorporate membrane active peptides into DNA complexes in order to improve their transfer from the endosome into the cytoplasm and their subsequent transport to the nucleus (see also peptide project).